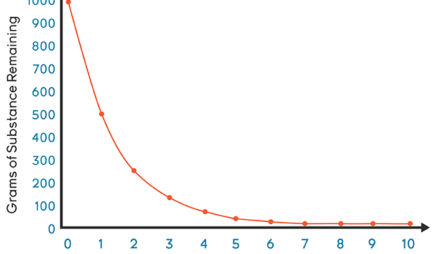
In Example 4 you found that ethyl chloride decomposes to ethylene and HCl in a first-order reaction that has a rate constant of 1.6 × 10 −6 s −1 at 650☌. Thus a first-order chemical reaction is 97% complete after 5 half-lives and 100% complete after 10 half-lives. The percent completion after 10 half-lives will be as follows: percent completion = ( 0.053 M − 5.2 × 10 − 5 M) ( 100 ) 0.053 M = 100 % Thus it takes almost 8 h for half of the cisplatin to hydrolyze.ī After 5 half-lives (about 38 h), the remaining concentration of cisplatin will be as follows: 0.053 M 2 5 = 0.053 M 32 = 0.0017 MĪfter 10 half-lives (77 h), the remaining concentration of cisplatin will be as follows: 0.053 M 2 10 = 0.053 M 1024 = 5.2 × 10 − 5 MĬ The percent completion after 5 half-lives will be as follows: percent completion = ( 0.053 M − 0.0017 M) ( 100 ) 0.053 = 97 % Then divide by the initial concentration, multiplying the fraction by 100 to obtain the percent completion.Ī We can calculate the half-life of the reaction using Equation 14.28: t 1 / 2 = 0.693 k = 0.693 1.5 × 10 − 3 min − 1 = 4.6 × 10 2 min Given: rate constant, initial concentration, and number of half-livesĪsked for: half-life, final concentrations, and percent completionĪ Use Equation 14.28 to calculate the half-life of the reaction.ī Multiply the initial concentration by 1/2 to the power corresponding to the number of half-lives to obtain the remaining concentrations after those half-lives.Ĭ Subtract the remaining concentration from the initial concentration. If a freshly prepared solution of cisplatin has a concentration of 0.053 M, what will be the concentration of cisplatin after 5 half-lives? after 10 half-lives? What is the percent completion of the reaction after 5 half-lives? after 10 half-lives? Calculate the half-life for the hydrolysis reaction under these conditions. The anticancer drug cisplatin hydrolyzes in water with a rate constant of 1.5 × 10 −3 min −1 at pH 7.0 and 25☌. This becomes evident when we rearrange the integrated rate law for a first-order reaction ( Equation 14.21) to produce the following equation: The half-life of a first-order reaction is independent of the concentration of the reactants. This is not true for zeroth- and second-order reactions. The half-life of a first-order reaction under a given set of reaction conditions is a constant. If two reactions have the same order, the faster reaction will have a shorter half-life, and the slower reaction will have a longer half-life. Thus the half-life of a reaction is the time required for the reactant concentration to decrease from 0 to 0/2. This period of time is called the half-life The period of time it takes for the concentration of a reactant to decrease to one-half its initial value. In addition, tritium dating can be useful in determining the age of wines and brandies.Another approach to describing reaction rates is based on the time required for the concentration of a reactant to decrease to one-half its initial value. Chemists and geologists use tritium dating to determine the age of water (ocean and fresh). For example, tritium (t 1/2 =12.3 years) can date samples within an age range of 1-100 years. Isotopes with shorter half-lives are used to date more recent samples. This radioactivity approach can be used to detecting fake wine vintages too. Present day estimates for the age of the Earth's crust from this method is at 4 billion years.

A useful concept is half-life (symbol is \(t_\)-206 in a sample of uranium mineral, the age of the mineral can be estimated. The decay process takes time and there is value in being able to express the rate at which a process occurs. During natural radioactive decay, not all atoms of an element are instantaneously changed to atoms of another element.
